Introduction
With the digitalization of manufacturing processes in the pharmaceutical and biotechnology sectors, the legal validity of electronic records and digital signatures has become a critical issue. 21 CFR Part 11, published by the U.S. Food and Drug Administration (FDA) in 1997, defines the conditions under which electronic records and electronic signatures are considered equivalent to their paper-based counterparts.
However, when this regulation was written, SCADA systems were very different from today. In traditional architectures, the development environment and the runtime environment were strictly separated: software was developed, compiled, tested, and deployed to production. This process naturally creates a "freeze point" and makes change management relatively straightforward.
But what if this separation disappears? In modern SCADA platforms, runtime and development coexist in the same environment; there is no compile-deploy cycle. While this offers tremendous flexibility for engineers, it raises an entirely new question for quality assurance managers.
In This Article: After summarizing the core requirements of FDA 21 CFR Part 11, we will discuss how modern SCADA architectures bring a different perspective to this regulation and what strategy quality assurance managers should follow in this new paradigm.
1. FDA 21 CFR Part 11: A Brief Overview
The regulation is built on three fundamental pillars:
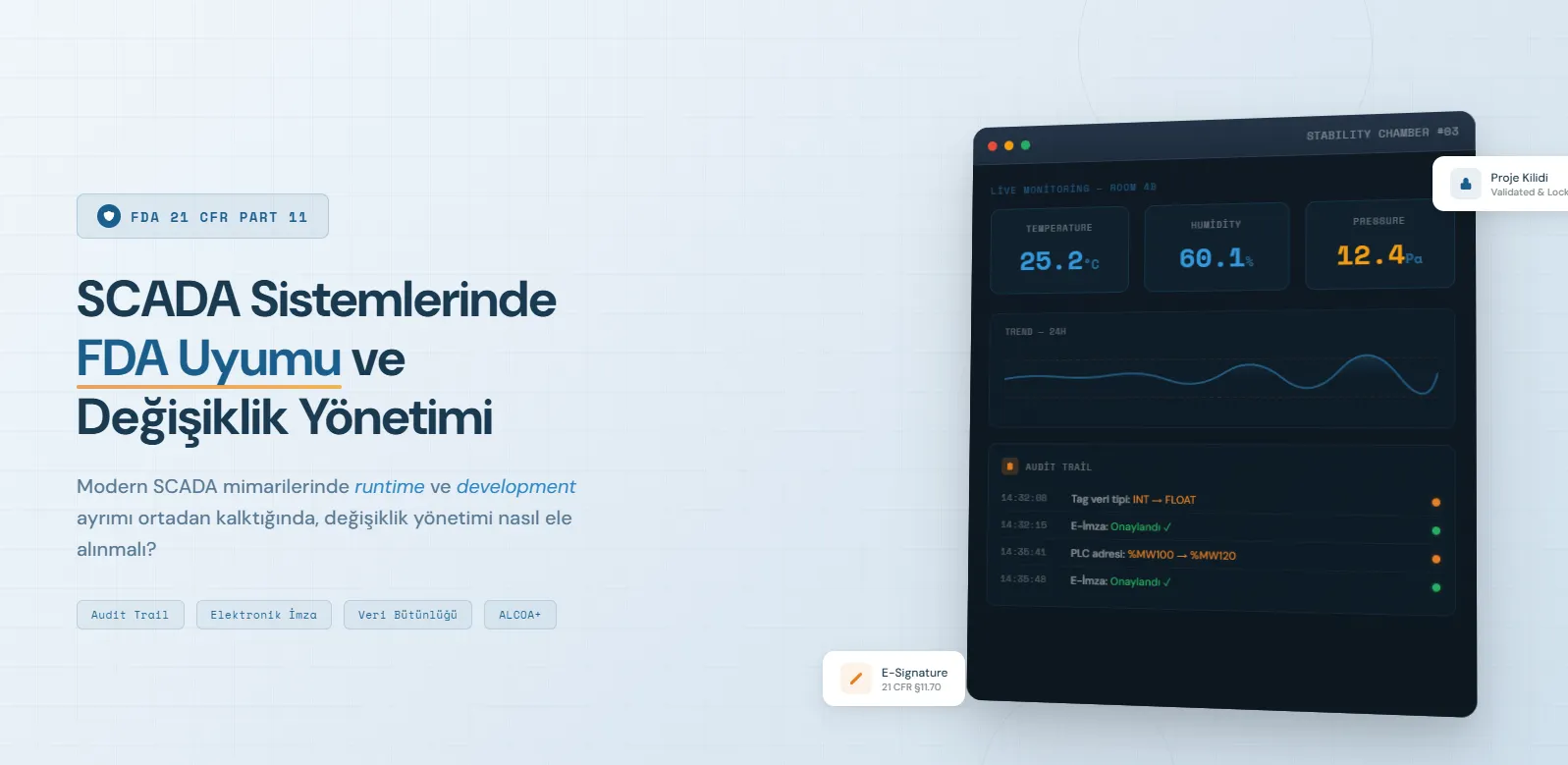
1.1 Audit Trail: Who, What, When, Why
The audit trail is the backbone of 21 CFR Part 11. It requires a chronological, immutable, and complete record of every change to process data. Each record must include:
- The user who made the change
- The old and new values of the modified data
- The timestamp of the operation (NTP-synchronized, UTC format)
- The reason for the change
1.2 ALCOA+ and Data Integrity
The FDA's data integrity framework, ALCOA+ principles, requires data to be Attributable, Legible, Contemporaneous, Original, Accurate, and additionally Complete, Consistent, Enduring, and Available. These principles apply not only to process data but also to system configuration data.
1.3 Electronic Signature Requirements
For electronic signatures to gain legal validity:
- Unique user identification
- Multi-factor authentication
- Cryptographic binding between signature and record
- Meaning indicated with each signature (approval, review, rejection)
2. Paradigm Shift: SCADA Without Compile-Deploy
Consider the traditional SCADA world: an engineer designs screens in the development environment, sets alarm limits, writes logic. Then this project is compiled, packaged, and loaded onto the production server. There is a natural "gate" in this process: the compilation moment.
In modern SCADA platforms, this gate does not exist. Runtime and development live in the same environment. When an engineer changes a tag's PLC address in the live system, this change takes effect immediately. No compilation, no deployment, no buffer zone in between.
Critical Difference
In traditional architectures, change management is naturally embedded in the compile-deploy process. In modern architectures, this process doesn't exist — therefore, the change management strategy must be consciously designed.
2.1 What Does This Flexibility Mean?
This architectural approach is a major advantage for field engineers. When a new sensor is added and the PLC program is updated, you can immediately configure the SCADA-side tag definition in the live system. Instant intervention, rapid adaptation, and uninterrupted operation become possible.
However, from the FDA 21 CFR Part 11 perspective, this flexibility raises an important question: Should structural changes made by a developer on the project fall within the scope of the audit trail?
3. Two Approaches: Digitalize or Lock?
There are two fundamental answers to this question, and both have strong arguments. The quality assurance manager's task is to choose the approach that best fits their facility's realities.
Approach A: Digitalize Developer Changes Too
In this approach, every configuration change made to the SCADA project is included in the FDA-compliant audit trail.
- Full traceability: Every change to the project is recorded with who made it, what changed, when it changed, and why it changed.
- Electronic signature integration: Approval via electronic signature can be mandated for critical configuration changes.
- Automatic version history: A timeline is created that allows returning to any point in the project.
- Continuous compliance: The risk of facing objections during audits that no changes were documented is eliminated.
Approach B: Test, Approve, Lock
In this approach, the traditional validation logic is maintained. Developer changes are made freely; the project is locked after passing the conventional IQ/OQ/PQ testing process.
- Free development: Engineers work on the project without any audit trail constraints.
- Validation process: The standard IQ/OQ/PQ testing process is applied when the project is completed.
- Project locking: The project is placed in a "frozen" state after validation.
- Change control: Any change to the locked project requires a formal change control process.
4. Comparative Evaluation
| Dimension | Approach A (Digitalize) | Approach B (Lock) |
|---|---|---|
| Development speed | Audit trail overhead possible | Maximum speed |
| Audit readiness | Always ready | Validation documentation needed |
| Traceability | Continuous and automatic | Post-validation only |
| Flexibility | Controlled flexibility | Restricted after locking |
| Implementation complexity | High (platform support needed) | Low (SOP-based) |
| Best for | Frequently changing, critical processes | Stable, rarely changing systems |
5. Hybrid Approach: Combining the Best of Both
In practice, the most suitable solution for most facilities will be a hybrid model that combines the strengths of both approaches.
Layered Change Management
Classify changes by criticality level:
| Level | Example | Requirement |
|---|---|---|
| Critical | Alarm limit, PLC address, control logic | Audit trail + E-signature + Approval |
| Medium | Screen layout, tag description | Audit trail + Automatic log |
| Low | Color scheme, language setting | Standard change record |
Development and Production Modes
- In development mode, changes are automatically logged but do not require approval.
- Transition to production mode requires a validation process and electronic signature.
- In production mode, an instant approval mechanism is activated for critical changes.
- Mode transitions are also included in the audit trail.
6. Practical Recommendations for Quality Assurance Managers
Regardless of which approach you choose, the following steps will strengthen your implementation process:
- Conduct a risk assessment: Identify which configuration elements in your SCADA project directly affect drug quality or patient safety.
- Define change categories: Classify critical structural elements and low-impact elements separately.
- Update your SOPs: Your existing SOPs are probably written based on compile-deploy assumptions. Create specific procedures for runtime development architecture.
- Work with your SCADA vendor: Evaluate the platform's audit trail, project locking, electronic signature, and role-based access control capabilities in detail.
- Create a training plan: Developers and operators must understand the requirements of the chosen approach and their own responsibilities.
- Plan periodic reviews: Regularly evaluate the chosen strategy and adapt it to evolving requirements.
Conclusion
When FDA 21 CFR Part 11 was written in 1997, SCADA systems had a strict compile-deploy cycle, and the regulation's assumptions were based on this reality. Today, the SCADA world has fundamentally changed. Modern platforms where runtime and development are intertwined offer flexibility and speed, while raising new questions about change management.
There is no single right answer to these questions. The right strategy depends on your facility's risk profile, the criticality of your production processes, your team's structure, and your existing quality management system. What matters is not ignoring this question, but making a conscious choice and documenting it.
Modern SCADA platforms have the infrastructure to technically support both approaches — and hybrid combinations of the two. As a quality assurance manager, your task is to align these technical capabilities with the right strategy and be able to say "We thought about this problem and here is our strategy" when facing an FDA auditor.